Humacyte, Inc (HUMA)
Stock Analysis Report
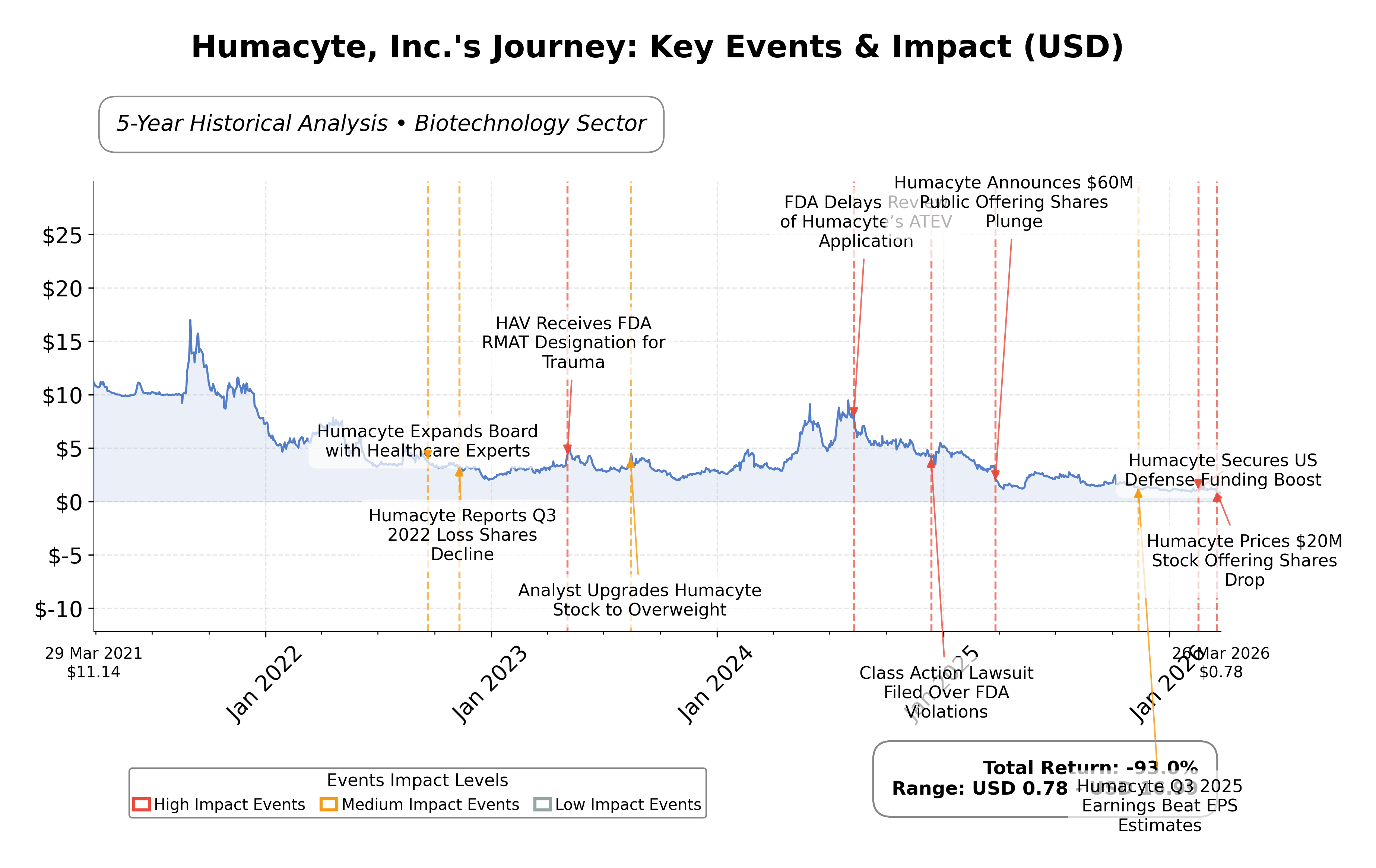
Stock Journey
Key Positives and Key Risks
Pros
- Innovative regenerative medicine technology with a gross margin of 65.5% indicating efficient product development.
- Strategic collaborations with the U.S. Department of Defense and international market expansion efforts, including a $1.475 million purchase commitment in Saudi Arabia.
- Recent $20 million registered direct offering enhances capital resources to support clinical and commercialization activities.
Cons
- Negative trailing P/E of -0.54 and forward P/E of -1.51 reflect ongoing losses and lack of profitability.
- Operating cash flow of -$105.5 million and free cash flow of -$74.2 million indicate significant cash burn.
- High debt level of approximately $50.97 million relative to negative shareholders’ equity raises financial leverage concerns.
Disclosure: This information is for general awareness and does not constitute investment advice
Report Summary
Humacyte, Inc. is a biotechnology company specializing in regenerative medicine, focusing on the development and commercialization of human acellular vessels (HAVs) designed to replace and repair damaged blood vessels. Operating within the healthcare and biopharmaceutical sectors, the company targets critical vascular conditions such as trauma and kidney dialysis, positioning itself as an innovator in tissue engineering and vascular health solutions. Headquartered in Durham, North Carolina, Humacyte aims to pioneer new medical technologies that offer alternatives to traditional biological tissues.
Financially, Humacyte reported a market capitalization of approximately $169.6 million with an enterprise value near $180.3 million. The company currently operates with negative earnings metrics, including a trailing P/E of -0.54 and a forward P/E of -1.51, reflecting ongoing losses. Revenue for the trailing twelve months stands at $1.57 million with a gross margin of 65.5%, while operating margins remain negative at -32.4%. Cash flow metrics indicate significant operating cash outflows, with a negative operating cash flow of approximately $105.5 million and free cash flow of about -$74.2 million, underscoring the companyâs investment phase.
From a strategic and technical perspective, Humacyte has recently submitted a Marketing Authorization Application for its product Symvess in Israel and secured a purchase commitment in Saudi Arabia, signaling international expansion efforts. Leadership remains stable with the founder and CEO actively involved, and the company has appointed a new SVP of business development. Key risks include high short interest (23.57% of shares outstanding) and substantial debt levels relative to equity, while strengths lie in its innovative product pipeline and partnerships with defense and military sectors.
Comparing Humacyte to regional biotechnology peers such as Vertex Pharmaceuticals and Regeneron Pharmaceuticals reveals significant valuation and scale disparities. Peers exhibit market caps in the tens of billions and positive P/E ratios, contrasting with Humacyteâs smaller size and negative earnings multiples. However, Humacyteâs return on equity of 2.82% slightly exceeds peersâ lower ROE figures, reflecting some operational efficiency despite losses. This peer context highlights the challenges and opportunities Humacyte faces as a smaller, early-stage biotech player within a competitive U.S. healthcare market.
Humacyte navigates a complex industry landscape marked by innovation demands, regulatory hurdles, and capital-intensive development cycles. Recent achievements include advancing clinical programs and expanding geographic reach, while ongoing challenges involve managing cash burn and meeting revenue targets. The company stands at a pivotal moment where successful commercialization and strategic partnerships could significantly enhance its market position, whereas continued financial pressures and competitive risks remain. Given these dynamics, a balanced perspective may be appropriate for those assessing the stock, emphasizing careful monitoring of operational milestones and market developments without immediate inclination toward accumulation or realization.
Company and Industry Overview
Company Basics
Price Performance
Company Size
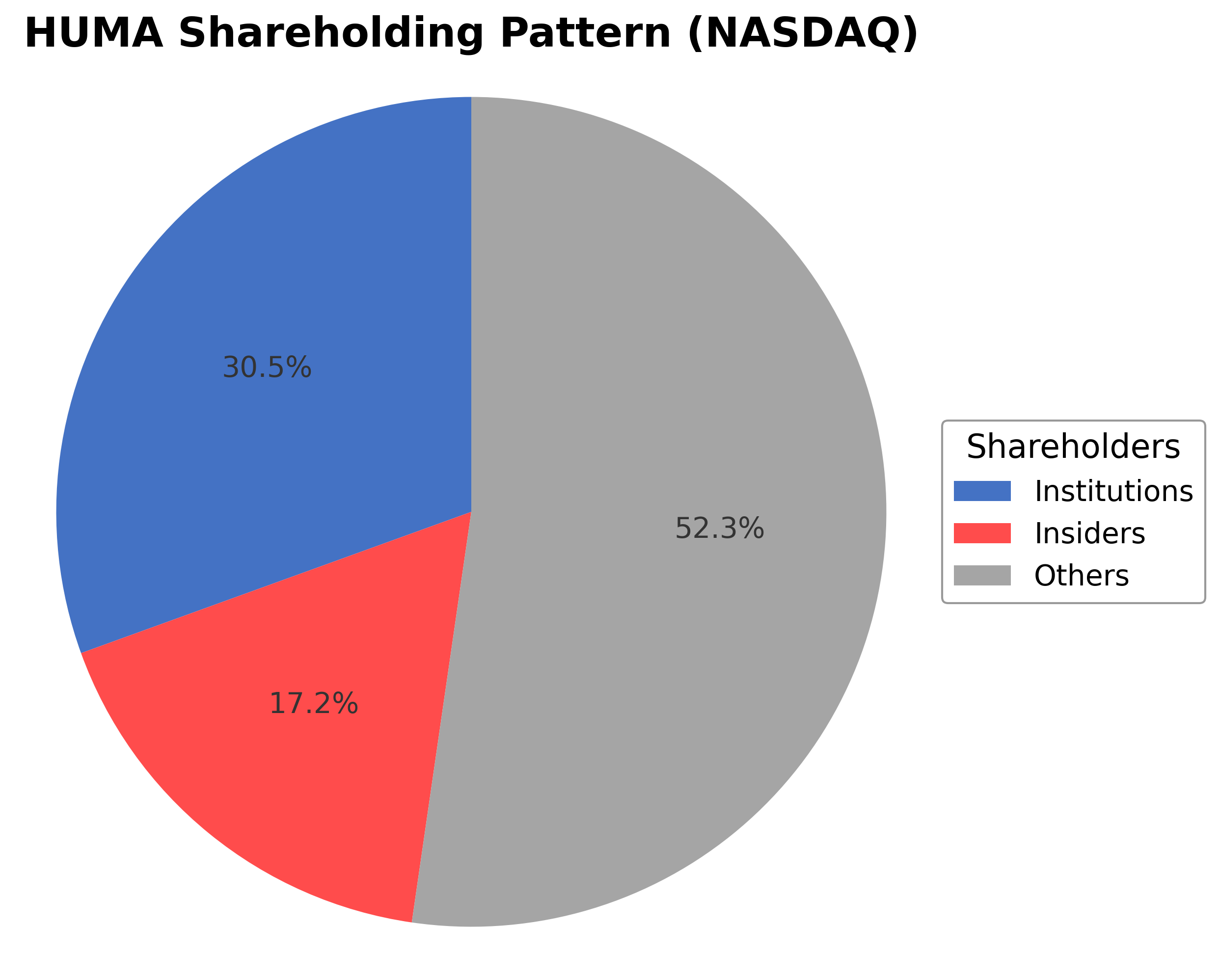
Humacyte, Inc.'s ownership structure comprises approximately 17.18% held by insiders including executives and board members, 30.53% by institutional investors such as mutual funds and asset managers, and the remaining 52.29% held by public shareholders including retail investors and employee stock plans. Over the past 12-24 months, institutional ownership has shown moderate activity with some accumulation by key funds, reflecting cautious interest in the company's regenerative medicine prospects. The shareholding pattern suggests a balanced market sentiment with significant institutional participation supporting governance and strategic initiatives. This ownership mix may influence future corporate actions, particularly in capital raising and strategic partnerships, within the competitive biotechnology sector.
Sector and Industry Analysis
Humacyte, Inc. (HUMA) operates within the Biotechnology sector, specifically focusing on bioengineered human tissues for therapeutic applications. The biotechnology sector is a significant and rapidly evolving segment of the broader healthcare industry, with a global market size exceeding hundreds of billions of dollars and projected to grow at a compound annual growth rate (CAGR) in the high single digits to low double digits over the next decade. Key players in this sector include large multinational firms such as Amgen, Gilead Sciences, and Regeneron, alongside numerous specialized biotech companies like Humacyte that focus on niche therapeutic areas and innovative biologics. The sector’s growth is driven by advances in molecular biology, genomics, and regenerative medicine, alongside increasing demand for novel treatments for chronic and rare diseases.
Within the tissue engineering and regenerative medicine industry, Humacyte’s proprietary platform for manufacturing human acellular vessels (HAVs) represents a cutting-edge approach targeting vascular repair and reconstruction. Industry trends emphasize the shift towards off-the-shelf, immunologically compatible biologics that reduce rejection risks and improve patient outcomes. There is growing interest in cell and gene therapies, personalized medicine, and the integration of bioengineered tissues with cellular therapies, such as pancreatic islet transplantation for diabetes. The convergence of biotechnology with advanced manufacturing techniques, including 3D bioprinting and scalable cell culture systems, is creating new opportunities for companies that can demonstrate clinical efficacy and regulatory approval.
The regulatory environment for biotechnology firms like Humacyte is complex and highly stringent, governed primarily by agencies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Key regulatory considerations include compliance with Good Manufacturing Practices (GMP), clinical trial protocols, and post-market surveillance. The pathway to approval for novel biologics and tissue-engineered products often involves extensive preclinical and clinical validation to demonstrate safety, efficacy, and immunogenicity profiles. Additionally, evolving policies around regenerative medicine and expedited approval pathways (e.g., FDA’s Breakthrough Therapy designation) can impact development timelines and market access. Regulatory uncertainty and reimbursement frameworks remain critical factors influencing commercial viability.
Competitive dynamics in the biotechnology and regenerative medicine industry are characterized by high barriers to entry due to substantial R&D investment requirements, intellectual property protections, and the need for specialized manufacturing capabilities. Market structure is fragmented, with a mix of large pharmaceutical companies, mid-sized biotechs, and emerging startups competing on innovation and clinical pipeline strength. Strategic partnerships, licensing agreements, and collaborations with academic institutions are common to accelerate development and commercialization. Companies like Humacyte that possess proprietary technology platforms and demonstrate clinical differentiation can achieve competitive positioning, but must navigate capital-intensive development cycles and commercialization challenges in a rapidly evolving landscape.
Note: Analysis synthesized from industry research, market reports, and regulatory filings. Information is subject to change based on market conditions.
Illustrative Scenario Analysis
DCF Assumptions:
Method: Two-Stage EPS-Priority Model
Financials
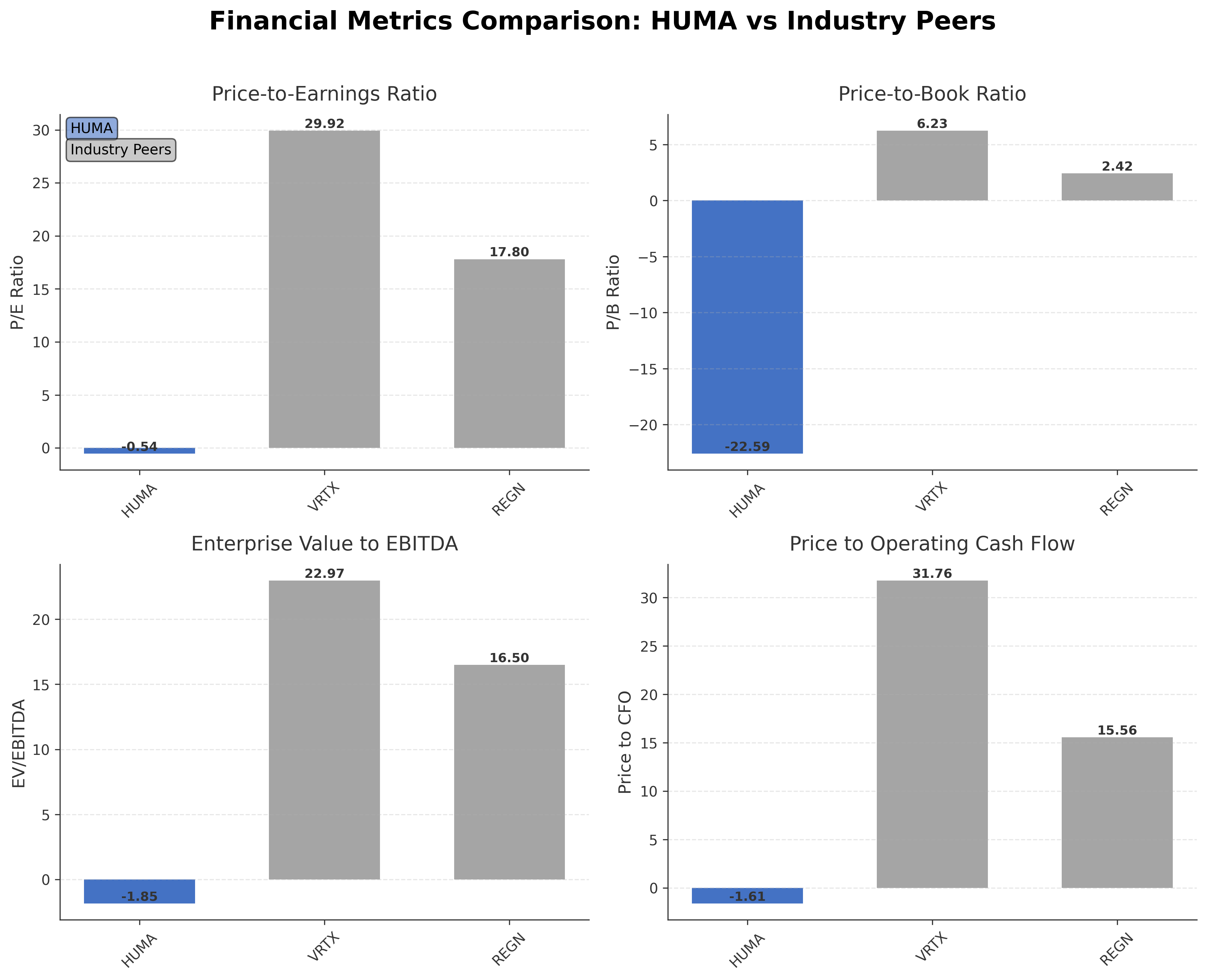
Peer Analysis
| Company Name | Market Cap | P/E Ratio | P/B Ratio | EV/EBITDA | Price to CFO |
|---|---|---|---|---|---|
| Humacyte, Inc. | $169.60M | -0.54 | -22.59 | -1.85 | -1.61 |
| Vertex Pharmaceuticals Incorporated | $115.32B | 29.92 | 6.23 | 22.97 | 31.76 |
| Regeneron Pharmaceuticals, Inc. | $77.48B | 17.80 | 2.42 | 16.50 | 15.56 |
Comparison Analysis: Humacyte, Inc. operates at a significantly smaller scale compared to its regional biotechnology peers Vertex Pharmaceuticals and Regeneron Pharmaceuticals, which have market capitalizations in the tens of billions. Humacyte's negative P/E and EV/EBITDA ratios reflect ongoing development-stage losses, contrasting with positive earnings multiples for its peers. The company's price-to-cash-flow ratio is also negative, indicating cash burn, whereas peers show positive cash flow metrics. Notably, Humacyte's return on equity of 2.82% surpasses the lower ROE figures of its larger peers, suggesting relatively better equity efficiency despite losses. This comparison underscores Humacyte's position as an early-stage innovator facing distinct financial and operational challenges within the competitive U.S. biotech industry.
Financial Metrics Comparison with Peers
Financial Statements
Comprehensive financial data including income, balance sheet, and cash flow metrics
Income Statement
| fiscal_date | 2024-12-31 | 2023-12-31 | 2022-12-31 | 2021-12-31 | 2020-12-31 |
|---|---|---|---|---|---|
| Sales | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Operating Expense Research And Development | 88.60M | 76.55M | 63.26M | 61.34M | 54.08M |
| Operating Expense Selling General And Administrative | 25.80M | 20.39M | 22.88M | 21.13M | 12.01M |
| Operating Income | -114.40M | -96.94M | -84.58M | -81.21M | -64.60M |
| Non Operating Interest Income | 4.10M | 5.47M | 2.63M | 16.00K | 278.00K |
| Non Operating Interest Expense | 9.28M | 6.60M | 6.20M | 4.35M | 2.20M |
| Other Income Expense | -29.13M | -12.70M | 76.18M | 59.06M | N/A |
| Pretax Income | -148.70M | -110.78M | -11.96M | -26.48M | -66.52M |
| Net Income | -148.70M | -110.78M | -11.96M | -26.48M | -66.52M |
| Eps Basic | -1.26 | -1.07 | -0.12 | -0.66 | -0.65 |
| Eps Diluted | -1.26 | -1.07 | -0.12 | -0.66 | -0.65 |
| Basic Shares Outstanding | 118.48M | 103.42M | 103.05M | 39.97M | 103.00M |
| Diluted Shares Outstanding | 118.48M | 103.42M | 103.05M | 39.97M | 103.00M |
| Ebit | -139.42M | -104.18M | -5.76M | -22.13M | -64.32M |
| Ebitda | -103.10M | -83.75M | -73.74M | -72.95M | -55.97M |
| Net Income Continuous Operations | -148.70M | -110.78M | -11.96M | -26.48M | -66.52M |
| Operating Expense Other Operating Expenses | N/A | N/A | -1.56M | -1.26M | -1.49M |
Data provided by Twelve Data
Balance Sheet
| fiscal_date | 2024-12-31 | 2023-12-31 | 2022-12-31 | 2021-12-31 | 2020-12-31 |
|---|---|---|---|---|---|
| Cash And Cash Equivalents | 44.94M | 80.45M | 149.77M | 217.50M | 39.93M |
| Accounts Receivable | N/A | 0.00 | 31.00K | 176.00K | 113.00K |
| Total Assets | 137.87M | 128.22M | 204.30M | 286.53M | 106.69M |
| Total Liabilities | 190.54M | 114.68M | 87.37M | 164.36M | 457.00M |
| Long Term Debt | 13.62M | 16.29M | 39.19M | 49.15M | 24.64M |
| Shareholders Equity | -52.67M | 13.55M | 116.93M | 122.17M | -350.32M |
Data provided by Twelve Data
Cash Flow Statement
| fiscal_date | 2024-12-31 | 2023-12-31 | 2022-12-31 | 2021-12-31 | 2020-12-31 |
|---|---|---|---|---|---|
| Operating Activities Net Income | -148.70M | -110.78M | -11.96M | -26.48M | -66.52M |
| Operating Activities Depreciation | 7.19M | 7.72M | 8.21M | 8.24M | 8.35M |
| Operating Activities Stock Based Compensation | 6.14M | 6.83M | 6.18M | 10.15M | 4.69M |
| Operating Activities Other Non Cash Items | 40.85M | 13.65M | -74.18M | -54.74M | 103.00K |
| Operating Activities Accounts Receivable | 0.00 | 31.00K | 145.00K | -63.00K | 488.00K |
| Operating Activities Accounts Payable | -1.96M | 4.81M | -509.00K | -197.00K | -889.00K |
| Operating Activities Other Assets Liabilities | -53.00K | -50.00K | -218.00K | -158.00K | 207.00K |
| Operating Activities Operating Cash Flow | -96.54M | -77.78M | -72.33M | -63.25M | -53.57M |
| Investing Activities Capital Expenditures | -1.57M | -2.28M | -1.05M | -220.00K | -268.00K |
| Investing Activities Sale Of Investments | 0.00 | 2.11M | 16.00M | 0.00 | N/A |
| Investing Activities Investing Cash Flow | -1.57M | -173.00K | 4.84M | -8.22M | -268.00K |
| Financing Activities Long Term Debt Payments | -2.58M | -34.07M | -1.98M | -1.73M | -1.50M |
| Financing Activities Common Stock Issuance | 89.05M | N/A | N/A | N/A | N/A |
| Financing Activities Other Financing Charges | 19.39M | 38.01M | N/A | 238.46M | N/A |
| Financing Activities Financing Cash Flow | 105.86M | 3.94M | -1.98M | 266.38M | 1.75M |
| End Cash Position | 95.29M | 80.80M | 149.77M | 217.50M | 39.93M |
| Interest Paid | 0.00 | 1.61M | 2.67M | 1.12M | 0.00 |
| Free Cash Flow | -99.69M | -75.58M | -72.18M | -81.41M | -55.89M |
| Investing Activities Purchase Of Investments | N/A | 0.00 | -10.11M | -8.00M | 0.00 |
| Financing Activities Long Term Debt Issuance | N/A | N/A | 0.00 | 29.66M | 3.25M |
Data provided by Twelve Data
Technical Analysis
Key Insights
- The current trend shows a downward price movement with the stock trading near its 52-week low of $0.67, indicating bearish momentum.
- Key support levels are observed around $0.67, while resistance is noted near the 50-day moving average at approximately $1.08 and the 200-day moving average at $1.59.
- The stock price is below both the 50-day and 200-day moving averages, suggesting a sustained bearish trend in the medium to long term.
- Momentum indicators show an RSI below neutral levels, MACD remains negative, and the stochastic oscillator indicates oversold conditions, reflecting weak momentum but potential for short-term consolidation.
- Across daily, weekly, and monthly timeframes, the price action consistently demonstrates downward pressure with limited recovery attempts.
- Current technical setup suggests potential continuation of bearish scenarios unless significant volume or catalyst emerges to break resistance levels.
Trending News
1. Headline: Humacyte, Inc. (HUMA) Q4 2025 Earnings Call Transcript | Seeking Alpha
Summary: Humacyte, Inc. (HUMA) Q4 2025 Earnings Call March 27, 2026 8:00 AM EDTCompany ParticipantsLaura Niklason - Founder, President, CEO & DirectorDale Sander...
Sentiment: neutral
2. Headline: Earnings call transcript: Humacyte Q4 2025 revenue misses forecast By Investing.com
Summary: Humacyte Inc. reported its fourth-quarter 2025 earnings, revealing that both its earnings per share (EPS) and revenue were in line with expectations but fell short in revenue forecasts. The company reported an EPS of -$0.13, matching analyst forecasts, while revenue reached $500,000, significantly ...
Sentiment: negative
3. Headline: Humacyte Inc reports results for the quarter ended December 31 - Earnings Summary — TradingView News
Summary: FORECAST CHANGESRECOMMENDATIONSThis summary was machine generated from LSEG data March 27 at 12:22 p.m. UTC. All figures in US dollars unless otherwise stated. (For questions concerning the data in this report, contact Estimates.Support@lseg.com. For any other questions or feedback, contact ...
Sentiment: neutral
4. Headline: Humacyte Announces Fourth Quarter and Year End 2025 Financial Results and Provides Business Update
Summary: New U.S. Department of Defense ... and incorporation of biologic vascular repair technologies for the warfighters suffering from traumatic vascular injuries. The Company is working with leaders in the military and the Pentagon to ensure that American service personnel will have access to Symvess. Submitted Marketing Authorization Application for Symvess in Israel: In March 2026 Humacyte submitted ...
Sentiment: neutral
5. Headline: Humacyte misses revenue target despite in-line earnings By Investing.com
Summary: DURHAM, N.C. - On Friday, Humacyte, Inc. (NASDAQ:HUMA) reported fourth quarter results that fell short of revenue expectations, though adjusted earnings per share matched analyst estimates.
Sentiment: negative
6. Headline: Humacyte posts $2M 2025 revenue, eyes June Phase 3 data | HUMA Stock News
Summary: New U.S. Department of Defense ... and incorporation of biologic vascular repair technologies for the warfighters suffering from traumatic vascular injuries. The Company is working with leaders in the military and the Pentagon to ensure that American service personnel will have access to Symvess. Submitted Marketing Authorization Application for Symvess in Israel: In March 2026 Humacyte submitted ...
Sentiment: neutral
7. Headline: Humacyte appoints Rick McElheny SVP of business development | HUMA Stock News
Summary: Motorola Solutions Canada Networks Inc. to Acquire Bell Canada’s ... ... Unity Releases Preliminary First Quarter Results Exceeding Guidan... ... Xanadu Quantum Technologies Limited Announces Closing of Business... ... Faraday Future Completes Delivery of Additional EAI Robots to New... ... Humacyte ...
Sentiment: neutral
8. Headline: Is Humacyte (HUMA) Using Middle East Traction And Equity Issuance To Redefine Its Risk Profile? - Simply Wall St News
Summary: Humacyte, Inc. recently secured a binding minimum purchase commitment of US$1.475 million for its Symvess acellular tissue engineered vessel in Saudi Arabia, alongside filing a US$20 million follow-on common stock offering and submitting a Marketing Authorization Application for Symvess in ...
Sentiment: positive
9. Headline: Humacyte Announces $20 Million Registered Direct Offering - TipRanks.com
Summary: The latest announcement is out from Humacyte ( ($HUMA) ). On March 19, 2026, Humacyte, Inc. entered into securities purchase agreements for a registered direct offe...
Sentiment: neutral
10. Headline: Humacyte Secures $1.5M Saudi Order – Why Did HUMA Stock Crash To Record Low Today?
Summary: Shares of Humacyte, Inc.
Sentiment: negative
Powered by Brave
Recent Updates
News Summary
Recent news coverage of Humacyte, Inc. highlights the company's Q4 2025 financial results, which showed a loss per share in line with estimates but revenue below expectations. The company continues to advance its Symvess product internationally, securing a $1.475 million purchase commitment in Saudi Arabia and submitting a Marketing Authorization Application in Israel. Leadership updates include the appointment of a new Senior Vice President of Business Development, aimed at bolstering commercial efforts. Additionally, Humacyte completed a $20 million registered direct offering to support its clinical and commercialization activities. Market analysts have adjusted price targets reflecting a cautious outlook amid lower sales forecasts, while strategic collaborations with the U.S. Department of Defense underscore ongoing efforts to expand product applications. Overall, these developments indicate a focus on capitalizing growth opportunities while managing operational challenges in a competitive biotech environment.
News Sentiment
Sentiment across recent updates is predominantly neutral with some negative tones related to missed revenue targets. Positive aspects include strategic partnerships and capital raises, which contribute to a balanced market perception. The mixed sentiment reflects the company's transitional phase from development to commercialization, with cautious optimism tempered by financial performance pressures.
Analytical Overview
Analysis Summary
Humacyte’s valuation metrics, including a negative trailing P/E of -0.54 and forward P/E of -1.51, contrast sharply with industry averages that typically show positive earnings multiples, reflecting the company’s developmental stage and ongoing losses. The price-to-sales and price-to-book ratios are notably high in magnitude but negative, indicating valuation challenges relative to revenue and book value.
The company’s growth trajectory shows modest quarterly revenue growth of approximately 1.5%, but overall revenue remains low at $1.57 million TTM, with significant negative operating cash flow of $105.5 million, highlighting substantial investment in R&D and commercialization efforts.
Financial health indicators reveal a current ratio of 1.62, suggesting adequate short-term liquidity, but total debt of $50.97 million against negative equity points to leverage concerns. Negative operating and free cash flows emphasize ongoing cash burn, necessitating capital raises such as the recent $20 million offering.
Sector-specific challenges include high regulatory barriers, long product development cycles, and competitive pressures in regenerative medicine. Opportunities arise from strategic partnerships, military collaborations, and international market expansion, which may support future revenue growth.
Considering the U.S. biotechnology market environment, regulatory complexities and innovation demands shape Humacyte’s operational context. The company’s positioning in regenerative medicine offers potential competitive advantages, though execution risks remain significant.
Investment Conclusion
Supporting Factors: Primary supporting factors include Humacyte’s innovative product pipeline in regenerative medicine, strategic collaborations with defense sectors, and recent capital infusion to support development. Main risk factors to monitor are ongoing negative cash flows, high leverage relative to equity, and revenue shortfalls against forecasts. The appropriate investment timeframe aligns with medium to long-term horizons given the company’s development stage and commercialization progress. Overall, the analysis reflects a balanced outlook with both growth potential and financial challenges.
Risk Factors: No data
SWOT Analysis
Strengths
- The company specializes in innovative regenerative medicine technology with human acellular vessels.
- Strong strategic collaborations with the U.S. Department of Defense enhance market positioning.
- Recent capital raise of $20 million supports ongoing clinical and commercialization efforts.
- Return on equity of 2.82% indicates operational efficiency despite losses.
Weaknesses
- Negative earnings and cash flow metrics reflect ongoing financial losses.
- High debt levels relative to equity raise leverage concerns.
- Revenue remains low with recent quarters missing sales forecasts.
- Significant short interest at 23.57% of shares outstanding indicates market skepticism.
Opportunities
- International expansion through Marketing Authorization Application submissions and purchase commitments.
- Growing demand for vascular repair solutions in trauma and dialysis markets.
- Potential for increased adoption in military and defense healthcare sectors.
- Advancements in tissue engineering could open new therapeutic applications.
Threats
- Regulatory hurdles and lengthy approval processes may delay commercialization.
- Competitive pressures from larger biotechnology firms with greater resources.
- Market volatility and investor sentiment influenced by financial performance.
- Dependence on successful clinical trial outcomes for future growth.
Company Description
Humacyte Inc. is a biotechnology company specializing in regenerative medicine. The company primarily aims to transform medicine through the development and commercialization of human acellular vessels (HAVs), which are bioengineered tissues designed to replace and repair damaged human blood vessels. Humacyte's technology addresses significant challenges in vascular health, often targeting critical conditions such as vascular trauma and kidney dialysis. Operating within the healthcare and biopharmaceutical sectors, the company contributes to advancing medical solutions by fostering innovative approaches to tissue regeneration. Headquartered in Durham, North Carolina, Humacyte plays a crucial role in pioneering new pathways in regenerative medicine, seeking to improve outcomes for patients requiring vascular replacements. As part of the market, it represents a key player in developing cutting-edge medical technologies that potentially offer viable alternatives to traditional biological tissue.